|
This moderate redistribution suggests changes in the interplay between active and passive perfusion redistribution mechanisms (including hypoxic vasoconstriction and gravitational effects), not the appearance of thromboembolism. In this early ARDS model, increases in alveolar dead-space occur within 20 hours due to the regional redistribution of perfusion and not ventilation. The increased alveolar dead-space derived mostly from areas with intermediate V ˙ / Q ˙ (0.5≤ V ˙ / Q ˙ ≤10), not areas of nearly "complete" dead-space ( V ˙ / Q ˙ >10). 031), while the spatial distribution of ventilation did not change, resulting in increased V ˙ / Q ˙ in non-dependent regions. This was mediated by perfusion redistribution away from non-dependent regions (-5.9%, P =. 031), mainly in non-dependent regions (+0.03, P =. 
Maps of voxel-level lung ventilation, perfusion, V ˙ / Q ˙, CO 2 partial pressures, and alveolar dead-space fraction were estimated from positron emission tomography at baseline and 20 hours.Īlveolar dead-space fraction increased during the 20 hours (+0.05, P =. 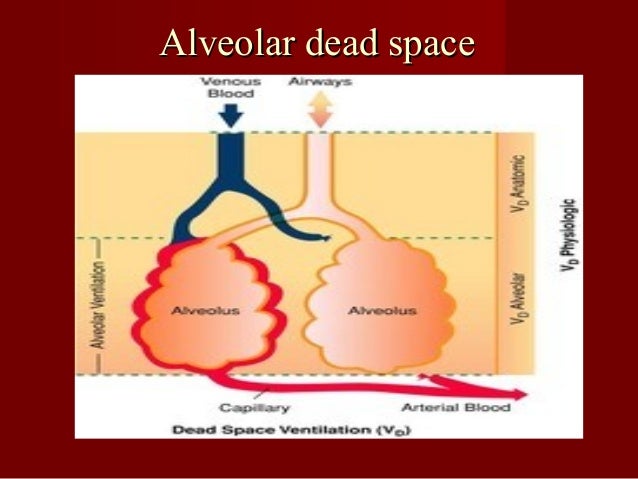
We hypothesized that: increased dead-space results from the spatial redistribution of pulmonary perfusion, not ventilation such redistribution is not related to thromboembolism (ie, areas with perfusion = 0 and infinite ventilation-perfusion ratio, V ˙ / Q ˙ ), but rather to moderate shifts of perfusion increasing V ˙ / Q ˙ in non-dependent regions.įive healthy anesthetized sheep received protective ventilation for 20 hours, while endotoxin was continuously infused. We aimed to investigate the physiological mechanism and spatial distribution of increased physiological dead-space, an early marker of ARDS mortality, in the initial stages of ARDS.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
